📖 Class 10 Science Previous Year Questions (PYQs)
🎯 Board Exam Prep • CBSE / ICSE / State Boards
⭐ Real previous year board exam MCQs — Test your knowledge!
👉 Click "Show Answer & Explanation" on any question to reveal the correct answer along with a detailed concept explanation.
Perfect for self-assessment and revision.
✅ Interactive MCQ | Answer Reveal
📚 PYQs with Explanations
📝 Chapter-wise & Mixed Board Exam Questions
Below are previous year board exam questions for Class 10 Science (Physics, Chemistry, Biology). Each question includes 4 options. Click the button to see the correct answer + concept explanation.
📌 Q1 (Board PYQ)
Electrolysis of water is a decomposition reaction. The mass ratio (MH : MO) of hydrogen and oxygen gases liberated at the electrodes during electrolysis of water is: (1 Mark)
✅ Correct Answer: D
During the electrolysis of water, water decomposes to form hydrogen and oxygen gases: The volume ratio of hydrogen to oxygen is 2 : 1, but since oxygen is eight times heavier than hydrogen, the mass ratio (Mₕ : Mₒ) is 1 : 8.
During the electrolysis of water, water decomposes to form hydrogen and oxygen gases: The volume ratio of hydrogen to oxygen is 2 : 1, but since oxygen is eight times heavier than hydrogen, the mass ratio (Mₕ : Mₒ) is 1 : 8.
📌 Q2 (Board PYQ - )
Consider the following reactions: (1 Mark)
- Dilute hydrochloric acid reacts with sodium hydroxide
- Magnesium oxide reacts with dilute hydrochloric acid
- Carbon dioxide reacts with sodium hydroxide
✅ Correct Answer: A (Salt and water is formed)
In all three reactions, an acid reacts with a base (or basic oxide) to produce salt and water, which is a neutralisation reaction — a type of double displacement reaction.
In all three reactions, an acid reacts with a base (or basic oxide) to produce salt and water, which is a neutralisation reaction — a type of double displacement reaction.
📌 Q3
In which one of the following situations a chemical reaction does not occur? (1 Mark)
✅ Correct Answer: D (Melting of glaciers)
Melting of glaciers is only a physical change — the state of water changes from solid (ice) to liquid, but no new substance is formed. Hence, no chemical reaction takes place.
Melting of glaciers is only a physical change — the state of water changes from solid (ice) to liquid, but no new substance is formed. Hence, no chemical reaction takes place.
📌 Q4
The correct balanced chemical equation showing exothermic reaction in which natural gas burns in air is: (1 Mark)
✅ Correct Answer: D (CH₄ + 2O₂ → CO₂ + 2H₂O + Energy)
When natural gas (methane) burns in air, it combines with oxygen to form carbon dioxide and water, releasing heat energy. This makes it an exothermic reaction.
When natural gas (methane) burns in air, it combines with oxygen to form carbon dioxide and water, releasing heat energy. This makes it an exothermic reaction.
📌 Q5 (Previous Year - 2025)
Consider the following chemical equation: (1 Mark)
pAl + qH₂O → rAl₂O₃ + sH₂
To balance this equation, the values of ‘p’, ‘q’, ‘r’, and ‘s’ are:
pAl + qH₂O → rAl₂O₃ + sH₂
To balance this equation, the values of ‘p’, ‘q’, ‘r’, and ‘s’ are:
✅ Correct Answer: C (2, 3, 1, 3)
Write skeletal equation: Al + H₂O → Al₂O₃ + H₂ Balance Al → 2Al + H₂O → Al₂O₃ + H₂ Balance O → 3H₂O → Al₂O₃ + H₂ Balance H → 3H₂O → Al₂O₃ + 3H₂ Final: 2Al + 3H₂O → Al₂O₃ + 3H₂
Write skeletal equation: Al + H₂O → Al₂O₃ + H₂ Balance Al → 2Al + H₂O → Al₂O₃ + H₂ Balance O → 3H₂O → Al₂O₃ + H₂ Balance H → 3H₂O → Al₂O₃ + 3H₂ Final: 2Al + 3H₂O → Al₂O₃ + 3H₂
📌 Q6 (Board PYQ)
The main observations while burning magnesium ribbon in air are: (1 Mark)
- Magnesium ribbon burns with a dazzling white flame
- A white powder is formed
- Magnesium ribbon vaporises
- Aqueous solution of the white powder turns blue litmus red
✅ Correct Answer: C (1 and 2)
When magnesium ribbon burns in air, it burns with a dazzling white flame and forms a white powder of magnesium oxide, showing that a chemical reaction has taken place.
When magnesium ribbon burns in air, it burns with a dazzling white flame and forms a white powder of magnesium oxide, showing that a chemical reaction has taken place.
📌 Q7 (Board PYQ)
The values of a, b, c and d in the following balanced chemical equation are respectively: (1 Mark)
aPb(NO₃)₂ → bPbO + cNO₂ + dO₂
aPb(NO₃)₂ → bPbO + cNO₂ + dO₂
✅ Correct Answer: (D) 2, 2, 4, 1
Balancing Steps (1 Mark):
Balancing Steps (1 Mark):
- Write skeletal equation: Pb(NO₃)₂ → PbO + NO₂ + O₂.
- Balance Pb: 1 atom on each side → no change.
- Balance N: There are 2 nitrogen atoms in Pb(NO₃)₂, so write 2Pb(NO₃)₂ → 2PbO + 4NO₂ + O₂.
- Balance O: Left side has 12 oxygen atoms; right side has 2 (in PbO) + 8 (in NO₂) + 2 in O2 = 12 → balanced.
📌 Q8 (Board PYQ)
Examples of thermal decomposition reactions are: (1 Mark)
(i) 2AgCl → 2Ag + Cl₂
(ii) CaCO₃ → CaO + CO₂
(iii) 2H₂O → 2H₂ + O₂
(iv) 2KClO₃ → 2KCl + 3O₂
✅ Correct Answer: (D) 2 and 4
Thermal decomposition reactions are those that require heat to break down a compound into simpler substances. CaCO₃ → CaO + CO₂ (by heating) 2KClO₃ → 2KCl + 3O₂ (by heating) Both are thermal decomposition reactions.
Thermal decomposition reactions are those that require heat to break down a compound into simpler substances. CaCO₃ → CaO + CO₂ (by heating) 2KClO₃ → 2KCl + 3O₂ (by heating) Both are thermal decomposition reactions.
📌 Q9 (Board PYQ)
Assertion (A): Decomposition reactions are generally endothermic reactions.
Reason (R): Decomposition of organic matter into compost is an exothermic process. (1 Mark)
✅ Correct Answer: (B) Both A and R are true, but R is not the correct explanation of A
A: Decomposition reactions usually require energy (heat, light, or electricity), making them endothermic. R: Composting involves microbial breakdown, releasing heat (exothermic). Both are true, but R describes an exception (biological process), not a general explanation for A.
A: Decomposition reactions usually require energy (heat, light, or electricity), making them endothermic. R: Composting involves microbial breakdown, releasing heat (exothermic). Both are true, but R describes an exception (biological process), not a general explanation for A.
📌 Q10 (Board PYQ)
Assertion (A): Silver chloride turns grey in sunlight.
Reason (R): It decomposes into silver and chlorine in sunlight. (1 Mark)
✅ Correct Answer: (A) Both A and R are true and R is the correct explanation of A
When silver chloride is exposed to sunlight, it decomposes into silver and chlorine gas, turning the substance grey due to the formation of silver metal.
When silver chloride is exposed to sunlight, it decomposes into silver and chlorine gas, turning the substance grey due to the formation of silver metal.
📌 Q11 (Board PYQ)
Assertion (A): All exothermic reactions are accompanied with evolution of heat and light.
Reason (R): Combination reactions may or may not be exothermic. (1 Mark)
✅ Correct Answer: (D) A is false, but R is true
All exothermic reactions release heat, but not necessarily light. Also, combination reactions may or may not be exothermic — for example, formation of slaked lime is exothermic, but not all combination reactions release light.
All exothermic reactions release heat, but not necessarily light. Also, combination reactions may or may not be exothermic — for example, formation of slaked lime is exothermic, but not all combination reactions release light.
📌 Q12 (Board PYQ)
A student performs the following experiment in his school laboratory. List two observations to justify that in this experiment a chemical change has taken place. (2 Marks)
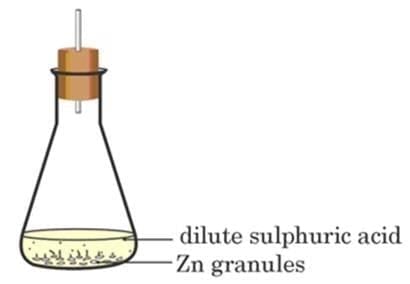

Bubbles of hydrogen gas are evolved when zinc granules react with dilute sulphuric acid.
The test tube becomes warm, showing that heat is produced — an exothermic chemical reaction has taken place.
Reaction:
Zn + H2SO4 → ZnSO4 + H2
📌 Q13 (Board PYQ)
Electrolysis of water is a decomposition reaction. The mass ratio (MH : MO) of hydrogen and oxygen gases liberated at the electrodes during electrolysis of water is: (1 Mark)
✅ Correct Answer: C (Rusting of iron)
Rusting of iron is a chemical change because a new substance (iron oxide) is formed, and the change is irreversible. Melting, dissolving, and breaking are physical changes.
Rusting of iron is a chemical change because a new substance (iron oxide) is formed, and the change is irreversible. Melting, dissolving, and breaking are physical changes.
📌 Q1 (Board PYQ)
Electrolysis of water is a decomposition reaction. The mass ratio (MH : MO) of hydrogen and oxygen gases liberated at the electrodes during electrolysis of water is: (1 Mark)
✅ Correct Answer: C (Rusting of iron)
Rusting of iron is a chemical change because a new substance (iron oxide) is formed, and the change is irreversible. Melting, dissolving, and breaking are physical changes.
Rusting of iron is a chemical change because a new substance (iron oxide) is formed, and the change is irreversible. Melting, dissolving, and breaking are physical changes.
📌 Q1 (Board PYQ)
Electrolysis of water is a decomposition reaction. The mass ratio (MH : MO) of hydrogen and oxygen gases liberated at the electrodes during electrolysis of water is: (1 Mark)
✅ Correct Answer: C (Rusting of iron)
Rusting of iron is a chemical change because a new substance (iron oxide) is formed, and the change is irreversible. Melting, dissolving, and breaking are physical changes.
Rusting of iron is a chemical change because a new substance (iron oxide) is formed, and the change is irreversible. Melting, dissolving, and breaking are physical changes.
📌 Q1 (Board PYQ)
Electrolysis of water is a decomposition reaction. The mass ratio (MH : MO) of hydrogen and oxygen gases liberated at the electrodes during electrolysis of water is: (1 Mark)
✅ Correct Answer: C (Rusting of iron)
Rusting of iron is a chemical change because a new substance (iron oxide) is formed, and the change is irreversible. Melting, dissolving, and breaking are physical changes.
Rusting of iron is a chemical change because a new substance (iron oxide) is formed, and the change is irreversible. Melting, dissolving, and breaking are physical changes.
📌 Q1 (Board PYQ)
Electrolysis of water is a decomposition reaction. The mass ratio (MH : MO) of hydrogen and oxygen gases liberated at the electrodes during electrolysis of water is: (1 Mark)
✅ Correct Answer: C (Rusting of iron)
Rusting of iron is a chemical change because a new substance (iron oxide) is formed, and the change is irreversible. Melting, dissolving, and breaking are physical changes.
Rusting of iron is a chemical change because a new substance (iron oxide) is formed, and the change is irreversible. Melting, dissolving, and breaking are physical changes.
📌 Q1 (Board PYQ)
Electrolysis of water is a decomposition reaction. The mass ratio (MH : MO) of hydrogen and oxygen gases liberated at the electrodes during electrolysis of water is: (1 Mark)
✅ Correct Answer: C (Rusting of iron)
Rusting of iron is a chemical change because a new substance (iron oxide) is formed, and the change is irreversible. Melting, dissolving, and breaking are physical changes.
Rusting of iron is a chemical change because a new substance (iron oxide) is formed, and the change is irreversible. Melting, dissolving, and breaking are physical changes.
📌 Q1 (Board PYQ)
Electrolysis of water is a decomposition reaction. The mass ratio (MH : MO) of hydrogen and oxygen gases liberated at the electrodes during electrolysis of water is: (1 Mark)
✅ Correct Answer: C (Rusting of iron)
Rusting of iron is a chemical change because a new substance (iron oxide) is formed, and the change is irreversible. Melting, dissolving, and breaking are physical changes.
Rusting of iron is a chemical change because a new substance (iron oxide) is formed, and the change is irreversible. Melting, dissolving, and breaking are physical changes.
📌 Q1 (Board PYQ)
Electrolysis of water is a decomposition reaction. The mass ratio (MH : MO) of hydrogen and oxygen gases liberated at the electrodes during electrolysis of water is: (1 Mark)
✅ Correct Answer: C (Rusting of iron)
Rusting of iron is a chemical change because a new substance (iron oxide) is formed, and the change is irreversible. Melting, dissolving, and breaking are physical changes.
Rusting of iron is a chemical change because a new substance (iron oxide) is formed, and the change is irreversible. Melting, dissolving, and breaking are physical changes.
